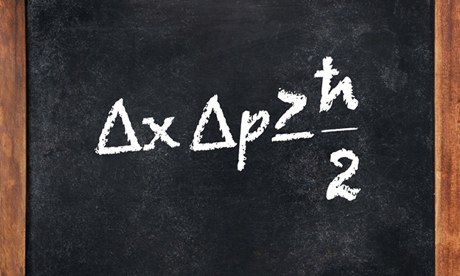Quantum mechanics is a framework which can be used to explain and predict various physical systems like nuclei, atoms and radiation or molecules. End of 19th century physics was dominated by classical or Newtonian mechanics, thermodynamics and electromagnetic theory. These theories were used to explain motion of material bodies, radiation studies, interaction of matter and radiation. At this point most of the known phenomenon were explained with these theories and classical theories had a great success.
The beginning of twentieth century brought few theories to light and challenged the ideas of classical mechanics. These ideas includes relativistic mechanics and Quantum mechanics. Classical theory failed to explain the motion of bodies traveling at speed very close to speed of light and it also failed to explain several microscopic phenomena like atomic stability, photo electric effect and blackbody radiation.
Max Plank was the biggest contributor to the beginning of quantum physics as he introduced the concept of quantum of energy to explain blackbody radiation. He postulated that the energy exchange between surrounding and radiation happens in discrete or quantized amounts. He explained that the energy exchange of electromagnetic waves of frequency with matter occurs only in integer multiples of
where h is planck’s constant. This idea lead to the solution of blackbody radiation and many other problems at that time.
Influenced by idea of Planck, Einstein postulated that this idea must be valid for light as well and he put forward the idea that light itself is made up off discrete bits of energy called photons and each photon has an energy of . Using this idea he beautifully explained photoelectric effect which has been a problem for physicist since 1887. Einstein also received Nobel Prize in Physics for this.
Neil Bohr in 1913 put forward the model of hydrogen atom by combining the concept of Photon, Planck’s quantum concept and Rutherford’s atomic model. His theory was that atoms can be found only in discrete states of energy and interaction of the atom with radiation takes place only in discrete amounts because of the transition of atom between its various discrete energy states.
Compton in 1923 with the help of x-ray scattering experiment conformed the corpuscular aspect of light and showed that x-ray photons behave like a particles with momenta . In 1924, de Broglie introduced an idea which associated electron with waves. He purposed that its not only wave that behaves like particle but the converse is also true and particles themselves behave like a wave and this was confirmed by Davisson and Garmer in 1927. By 1925 quantum mechanics was in full swing and scientist like Heisenberg and Schrodinger were in forefront of new discoveries and theories of Quantum physics.
The matrix mechanics and wave mechanics were two independent formulation of quantum mechanics. Matrix mechanics is used to describe atomic structure and was developed by Heisenberg. Heisenberg propounded his theory on the notion that the only allowed values of energy exchange between micro systems are those that are discrete quanta. Expressing quantities like angular momentum, momentum, position and energy in terms of matrices, he obtained an eigenvalue problem that describes the energy spectrum and the state vectors of the system.
The wave mechanics was formulated by Schrodinger and is more intuitive than matrix mechanics. Max Born proposed his probabilistic interpretation of wave mechanics. He took the square moduli of the wave functions that are solutions to the Schrodinger equation and the interpreted them as probability densities.
The concept of abstract objects like bras, kets and operators was introduced by Dirac.Dirac in 1928 derived an equation which describes the motion of electron. This equation predicted the existence of positron which is antiparticle of electron. This predicted particle was discovered in 1932 four years later.