Binding energy is the energy that holds a nucleus together and is equal to the mass defect of the nucleus. Nuclear binding energy is the energy that would be required to disassemble the nucleus of an atom into its component parts. These component parts are neutrons and protons, which are collectively called nucleons. Let us calculate the binding energy of an particle using the concept of mass defect
Mass of 2 protons + 2 neutrons =
= 4.031882
Mass of particle = 4.001506
Mass defect =
B.E. =
B.E. =
Binding energy per nucleon =
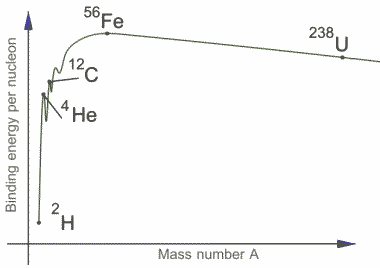
The binding energy per nucleon and the mass number is plotted in the figure. We can see that the curve rises steeply initially and then gradually reaches maximum of 8.79 MeV at A=56. This number A=56 corresponds to Iron . The curve now drops very slowly to somewhere around 7.6 MeV at the highest known mass number. From this graph we can say that nuclei of intermediate mass are more stable since high amount of energy is required to separate these nucleons. This also means that a large amount of energy will be liberated if heavier nuclei can be divided into lighter ones in nuclear fission.
 Packing fraction: The ratio between mass defect and the mass number is called the packing fraction. Packing fraction means mass defect per nucleon. Packing fraction is used to measure the comparative stability of the atom.
Packing fraction: The ratio between mass defect and the mass number is called the packing fraction. Packing fraction means mass defect per nucleon. Packing fraction is used to measure the comparative stability of the atom.
Packing fraction can be positive or negative. If packing fraction is negative, the isotopic mass is less than the mass number and in that case the mass gets transformed into energy in the formation of that nucleus according to the formula . This nuclei is more stable.
A positive packing fraction would mean a tendency towards instability. This is not essentially correct for atoms will lower atomic mass though. It should be noted that the element with mass numbers more than 230 are radioactive and goes through spontaneous disintegration.
Nuclear stability
The stability of nuclei are classified according to even and odd number of protons and neutrons
| Protons | Neutrons | Stable Nuclei |
|---|---|---|
| even | even | 160 |
| even | odd | 56 |
| odd | even | 52 |
| odd | odd | 4 |
For stable nuclei, we can see that nature prefers even number of protons and an even number of neutrons. The odd odd combination of proton and neutron in the nucleus are very less and are found only in lighter elements. For elements with large number of proton in the nucleus, the coulomb electrostatic force of repulsion becomes significant and the number of neutrons must be greater to compensate this repulsion effect.





Thanks to the author. It really helps in determining the concept of binding energy & stability of nucleus. I’ve a question.
Do u know How Raman effect differ from fluorescence and Rayleigh scattering?
Please send me the solution to harishjoshi401.hj@gmail.com
Thanks in advance!